Contract Researchers
ARM Company Customizations
Some of GDM's global corporate clients use customized ARM study definitions and validation lists. They also provide their customized study definitions to independent contract researchers. This approach ensures uniformity and enhanced data management.
Since independent contract researchers often conduct research studies for more than one company, GDM has a customized install for company customizations. (See below for installation instructions.)
Each company’s customizations are stored separately, so multiple companies’ customizations can reside on one ARM program. ARM automatically keeps separate settings for each customization, and refers to each as a “Settings profile.”
A contractor or cooperator may request sponsor customizations (pdf) within ARM. After approval, use the method described below in "Installation Directions" to install approved sponsor customizations.
Note: Always sent trials to sponsor using File - Sent to - External Sponsor/Cooperator, to ensure the sponsor receives all trial information.
Installation Directions
|
Opening Studies without Customized Company Files
A few research sponsors do not distribute their ARM customizations outside of their company. However, a contract researcher may occasionally receive a study created with customized study definitions that ARM cannot open because the study definitions are not available in standard ARM installations.
The message "Study definition 'xxxxxx.def' is not present in a known study definition directory. Can ARM search the drives on your computer to locate this study definition?" displays.
If not found, another message displays: "Study definition 'xxxxxx.def' not in current definition directory. Select the directory containing the definition next." Click the OK button.
If the company representative who sent the ARM study is not providing you with their customized study definitions, then on the "Select Study Definition" wizard choose: "Myself or another sponsor (standard GDMdef)":
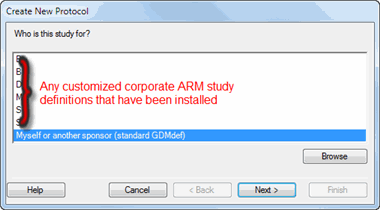
Click the Next button.
Choose G-All7 for most studies, or G-Seed7 for a pure variety testing study:
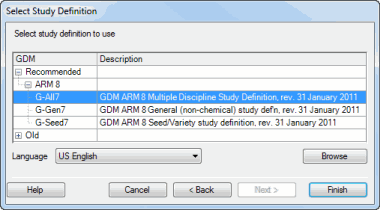
Click the Finish button.
Please ensure you are using the most recent ARM maintenance update, since many times the most recent update includes changes to improve compatibility when opening studies into a GDM study definition.
Instructions for Importing Study Files Using Standard GDM Study Definitions
- Save the ARM protocol *.edp or *.ede files into an ARM data directory, such as My Documents\ARM Data. Note the file names, since you will import them by file name in ARM.
- Start ARM, then select File - Import - EDE.
- Continue to step 4 if the files are included in the list of "Available Files" on the left of the ARM Select Batch dialog. Otherwise, change the drive (if needed) and directory on the right side of the ARM Select Batch dialog to the same directory where *.edp or *.ede file attachments were saved in step 1 above.
- Highlight an *.ede or *.edp file to import then click the Add button. Repeat this step for every file to be imported.
- Click the OK button once all the files are included in the "Selected Files" list on the ARM Select Batch dialog to begin the Import process.
- Select the NO button when asked whether to "Import into xxxxxx.def and xxxxxx.frm named in EDE file?" (where xxxxxx equals name of the company customized definitions.)
- When asked to "Enter name of protocol/trial file to create", you can assign the file name for the first ARM protocol or trial, and also the assign the directory to store each protocol or trial that is created.
- When asked to "Select study definition to use", you can identify the ARM study definition (*.def) file to use. For example, select G-All7.def for a crop protection study or a study where products are applied as a treatment factor, and select G-Seed7 for a seed variety testing study.
- When importing a trial *.ede file you are also asked to "Select site description format to use". Select the site description format (*.frm) name that matches the study definition name selected in previous step. You are not asked this question when importing a protocol *.edp file.
- ARM next imports each *.edp or *.ede file that was listed in the ARM Select Batch dialog.
- Click the Save button to save the study that ARM creates from the last EDP or EDE that was imported.
Sending Trial Results to Sponsor

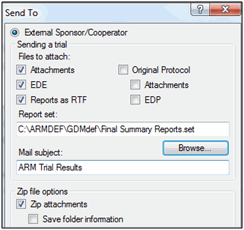
This will:
- Validate the trial
- Create an *.ede to import into sponsor study definitions
- Include the ARM trial so all original data is included
- Include attachments, providing all supplemental information
- Include reports printed to *.rtf for a quick overview of trial results

